Noe Rico Montanari et al., 2022
Multi-parametric analysis of livers reveals variation in intrahepatic inflammation of chronic hepatitis B infection
Relevant for
Pharma, CROs, Biotech
Background and aims: Chronic HBV is clinically defined in 4 phases by a combination of serum HBV DNA levels, HBeAg status and ALT: immunotolerant (IT), immune-active (IA), inactive carrier (IC) and HBeAg-negative hepatitis (ENEG). Immune and virological differences between phases as detected in blood have proven useful but are insufficient to appreciate the interrelation between the immune system and the virus since immune dynamics in blood and liver differ. Furthermore, the inflammatory response in the liver and parenchymal cells cannot be fully captured in blood.
Methods: Immunological composition and transcriptional profiles of FFPE core needle liver-biopsies in chronic HBV phases vs healthy were evaluated by multiplex immunofluorescence and RNA-Seq (n = 37 and 78, respectively).
Results: Irrespective of the phase-specific serological profiles, increased immune-gene expression and frequency was observed in chronic HBV compared to healthy livers. Greater transcriptomic de-regulation was seen in IA and ENEG (172 vs 243 DEGs) than in IT and IC (13 vs 35 DEGs) livers. ISG, immune-activation and exhaustion genes (ICOS, CTLA4, PDCD1) together with chemokine genes (CXCL10, CXCL9) were significantly induced in IA and ENEG livers. Moreover, distinct immune profiles associated with ALT elevation, and a more accentuated immune-exhaustion profile (CTLA4, TOX, SLAMF6, FOXP3) found in ENEG and set it apart from IA phase (LGALS9, PDCD1). Interestingly, all HBV phases showed downregulation of metabolic pathways vs healthy livers (fatty and bile acid metabolism). Finally, we found increased leukocyte infiltrate correlated with serum ALT, but not with HBV DNA or viral proteins.
Conclusion: Our comprehensive multi-parametric analysis of human livers revealed distinct inflammatory profiles and pronounced differences in intrahepatic gene profiles across all chronic HBV phases in comparison to healthy liver.
Summary: Immunological studies on chronic HBV remains largely restricted to assessment of peripheral responses due to the limited access to the site of infection, the liver. In this study, we comprehensively analyzed livers from a well-defined cohort of chronic HBV patients and uninfected controls with state-of-the-art techniques and evaluated the differences in gene expression profiles and inflammation characteristics across distinct disease phases of chronic HBV patients.
What is the problem?
Analyze inflammation characteristics across disease phases of chronic HBV patients versus uninfected controls
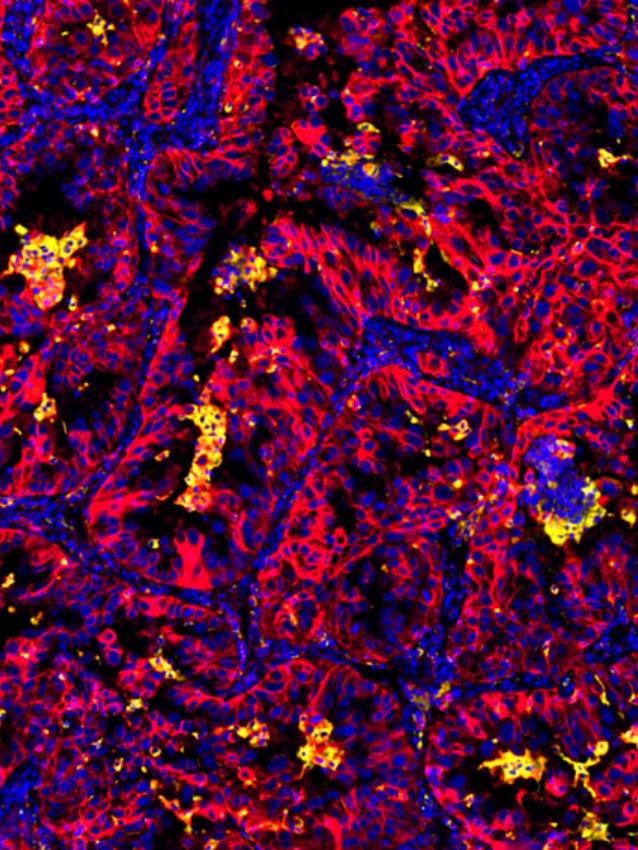
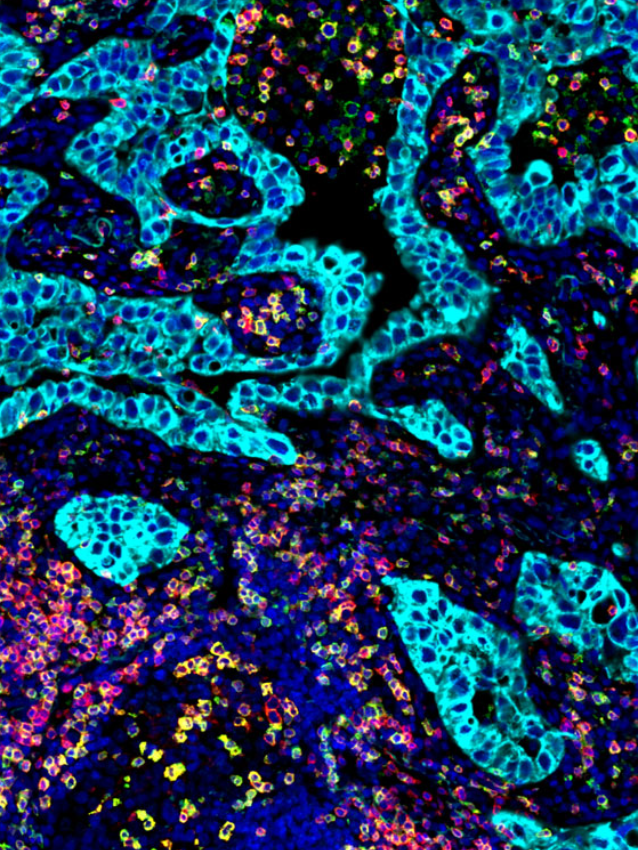
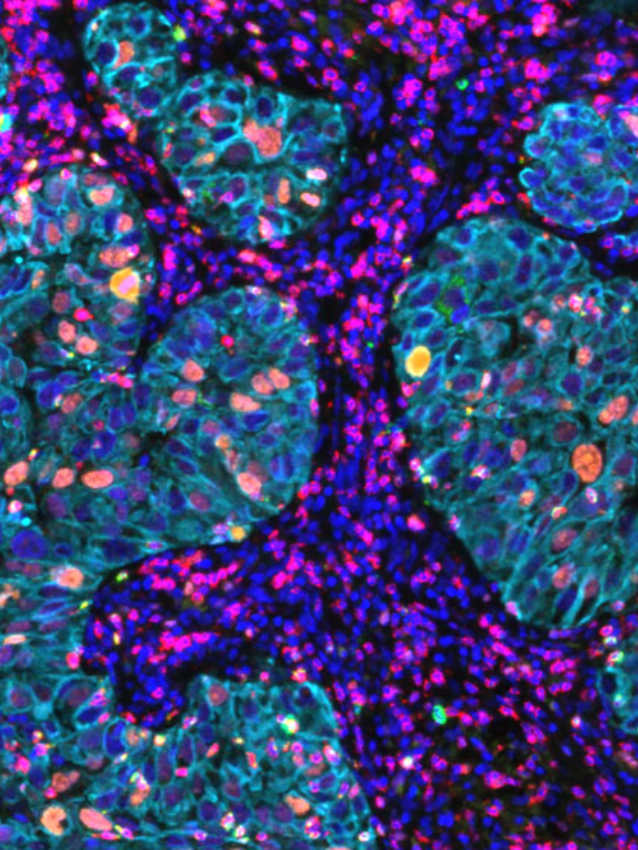
To date immunological studies on chronic HBV remains largely restricted to assessment of peripheral responses due to the limited access to the site of infection, the liver. In this study, the group wished to comprehensively analyze livers from a well-defined cohort of chronic HBV patients and uninfected controls with state-of-the-art techniques and evaluate the differences and inflammation characteristics across distinct disease phases of chronic HBV patients. Importantly, immunological composition was performed using whole tissue multiplex immunofluorescence (mIF) core needle liver biopsies from a unique cohort of uninfected healthy controls and chronic HBV patients. InSituPlex technology allows researchers to detect multiple analytes on a single FFPE slide.
Both RNASeq and multiplex immunofluorescence (mIF) was performed on liver biopsies to characterize the HBV clinical phases. Demonstrating that chronic HBV livers were characterized by higher immune-gene expression (i.e., ISGs, IRFs, T- and B-cell related) and leukocyte infiltrate (CD4+ and CD8+ T-cells and CD20+ B-cells) compared to healthy livers.
Results
The implementation of mIF provides a unique insight into cell subsets, specific phenotype co-localization and spatial biology
The group integrated the Ultivue InSituPlex® 12-plex Immunophenotyping Panel in their overall study design. The implementation of mIF provides a unique insight into cell subsets, specific phenotype co-localization and spatial insight with liver biopsies across all chronic HBV phases showing increased evidence of immune cell activity. The study highlighted the value of mIF affording co-localization and co-expression analysis. Further studies are warranted to better understand whether the limited but detectable T-cell infiltrate present in chronic infection and and IC phases affects intrahepatic responses and initiates cytopathic damage.