Back in the Saddle at the AACR
I’ve been a regular participant of the annual meeting of the American Association for Cancer Research (AACR) since 1998; it’s always been a great conference – an exciting time to learn about cutting edge innovations and research in cancer biology from talks and new technologies being showcased, to check out up to date content in posters (typically ahead of formal peer review, if being submitted for publication), and, most importantly to catch up with former colleagues. This year, after two long years of convening virtually, it was great to see so many unite in the city of New Orleans for #AACR2022.
April 28, 2022
In her opening remarks for the conference, Margaret Foti, CEO of the AACR, noted that “despite the challenges, this is an incredibly exciting, innovative, and hopeful time in cancer research. As reflected in the theme of this year’s meeting, decoding cancer complexity, integrating science, transforming patient outcomes, the pioneering strides you and your colleagues are making across the spectrum of cancer science are driving transformative progress against the disease at an exhilarating pace.” As we think about some of the strategies to deconvolute the complexities of cancer, especially within the tumor environment, one area in particular that dominated talks and posters this year was some of the recent advances in spatial technologies and multiplex approaches, from single to multiple biomarker assessment.
As outlined in his AACR 2022 summary, Colin Enderlein, DeciBio, observed that 2022 was “shaping up to be a transformational year for Multiplex Tissue Analysis (MTA) and Digital Pathology (DP), with ~20 clinical and marketing partnerships already announced these past ~4 months, and >2X the volume of spatial-omic activity seen at AACR vs prior years” (DeciBio). Indeed, a timely opinion piece from Nature Methods, published ahead of the AACR but referencing several areas of work highlighted specifically at the conference, had this to say about the use of multiplexed immunofluorescence (mIF); “we expect that advances in probes, barcoding strategies, affinity reagents, multiplexed detection, microscope sensitivity and microscope throughput will continue to move highly multiplexed tissue imaging forward in important ways. We also think improved methods for integrating multiplexed imaging data with other omics technologies will be crucial to the next phases of technological advancement. Finally, improved algorithmic and software tools for viewing and analyzing highly multiplexed and multi-modal omics data will also be needed to make the most of these advances.” I couldn’t agree more.
Ahead of the main conference, on Friday, April 8, in collaboration with Hanson Wade, Ultivue hosted our 2nd annual Biomarker Insights Summit, a day-long symposium designed to promote thought leadership and engage those working towards better understanding, diagnosis, and treatment of cancer via immuno-oncology approaches. Our esteemed speakers and panelists shared their efforts and lessons learned to better inform therapeutic approaches, including how innovative multiplex and digital pathology insights can provide benefit to clinical trials. The lively morning session covered a variety of “spatial phenomics” techniques to better understand tumor diversity, and why a greater understanding of the immune repertoire within the tumor microenvironment will be necessary to better stratify patients. Importantly, while our panel discussion centered on how we strategically think beyond the clinical utility of PD-L1, it’s clear that there remain differences between views of the pathologists vs. oncologists around the most optimal approach to clinical use. A networking lunch was extremely valuable for candid discussions, and it was great to see such enthusiasm for integration of spatial analysis into translational programs, including the variety of workflows available. The afternoon session continued with many details of implementing mIF, including assay reproducibility and validation, image analysis methods and preferred parameters to describe tumor and immune cells within a given tissue. The dynamic panel discussion touched on sources of ground truth for training AI models, a concern given the inter-pathologist variation in tumor assessments. With additional talks from our co-marketing partners at Aignostics and Paige, it was encouraging to see the emergence of cohesive spatial workflows that combine the power of mIF and AI development, a topic we look forward to hearing more about in the future.
Artificial intelligence (AI), which holds great potential to improve cancer diagnosis, prognosis, and prediction of response to therapy, was featured widely at AACR 2022. Dr. Su-In Lee, University of Washington, in a session on AI models discussed how explainable AI (XAI) can be used to predict clinical outcomes. She showed how datasets from patient tumor cohorts that include gene expression levels and the effects of different combinations of drugs on their biological targets can be used to train a machine learning model that can predict how synergistic untested combinations of drugs might act in different patients. “XAI can help us make new biological discoveries from data and may inform clinical decisions, and even open new research directions in biomedicine,” she said. Many speakers in the session, however, cautioned that while promising, such techniques have not yet become standard practice because of a lack of interpretability and transferability in translational medicine.
In keeping with the theme of integrating biomarker discovery with digital pathology, an important educational session from Drs. David Rimm, Janis Taube, and Tomas Kirchhoff discussed the importance of predictive biomarkers in the adjuvant immunotherapy setting, especially biomarkers to determine which patients need additional therapy and who can stop treatment. It was clear that the current crop of tissue-based biomarkers of immune checkpoint inhibitor (ICI) outcomes have severe limitations in precision medicine applications, owing to their woefully inadequate predictive power, high assessment costs, and difficulties with specimen availability. However, as noted by Dr. Taube, analysis of the resected specimens that have already been under therapeutic pressure can provide valuable information on how well the immunotherapy has worked in that patient, as well as revealing mechanisms of recurrence and resistance.
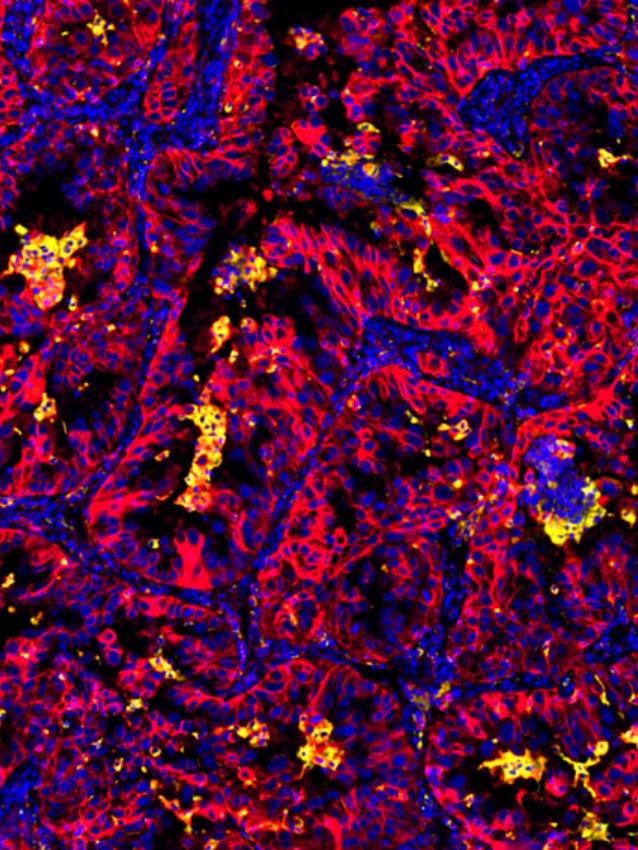
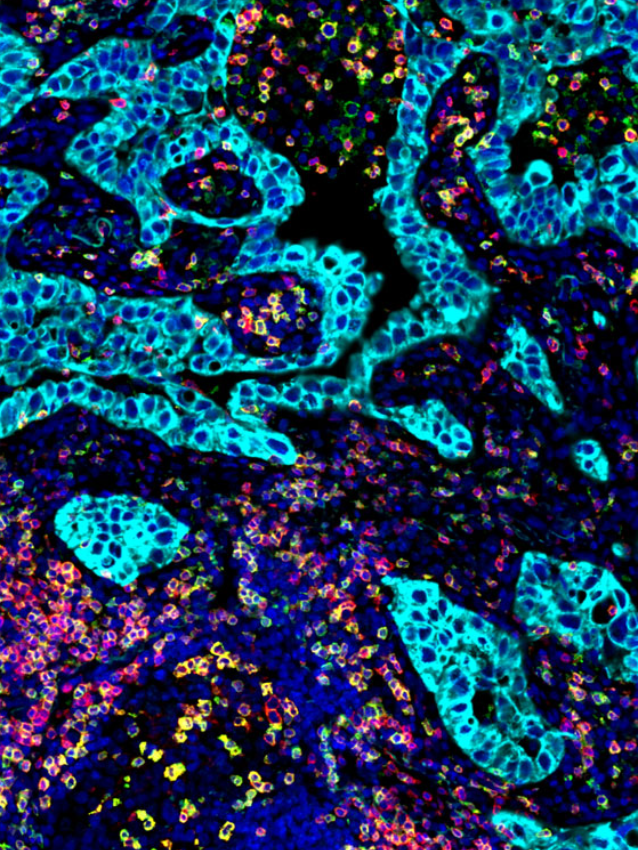
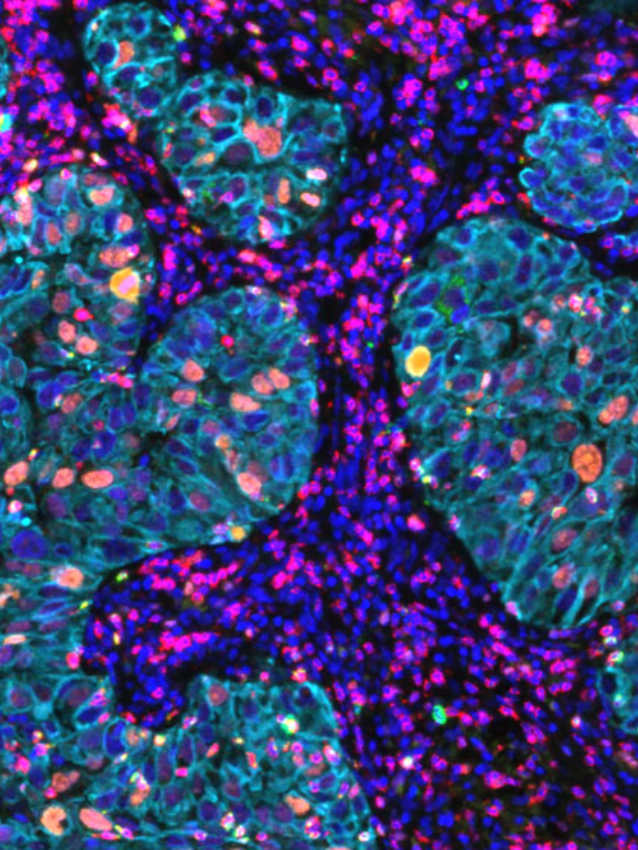
Ultivue presented several posters during the conference sessions, including a pre-optimized protocol for mIF staining on mouse tissue that provides a new way to investigate T-cell phenotypes in preclinical syngeneic or genetically modified mouse model tissues. Another poster featured our new flexible mIF assays (FlexVUE™ panels) for discerning relevant distributions of infiltrating immune cells in tumors coupled with UltiStacker® software for producing detailed spatial characterizations of specific cell phenotypes. Many scientists who stopped by our posters commented on, “how beautiful our images are.” A collaborative poster between Ultivue, Fluidigm (recently rebranded as Standard Bio Tools), and Visiopharm combined each company’s expertise to create a workflow that can unravel the complexity of the tumor microenvironment in pancreatic adenocarcinoma. Our newest workflow demonstrated co-detection of both protein and RNA on a single tissue section, and we look forward to sharing a white paper on this exciting application in the next few weeks.
Overall, despite the long days and my inappropriate footwear (I should have known that a conference was not the time to rock new heels), it was a fantastic educational meeting, and so great to hear from so many peers and experts at the forefront of cancer research. It was great to talk to the many vendors working on their unique solutions in the field of spatial biology, and especially to the scientists using these methods to unravel the complexity of cancer. It is becoming increasingly clear that the integration of “spatial phenomics” into translational efforts in the lab and the clinic is poised to deliver a much-needed promise to patients of better outcomes. However, a key takeaway is that the fight is not over yet; we must work better together!